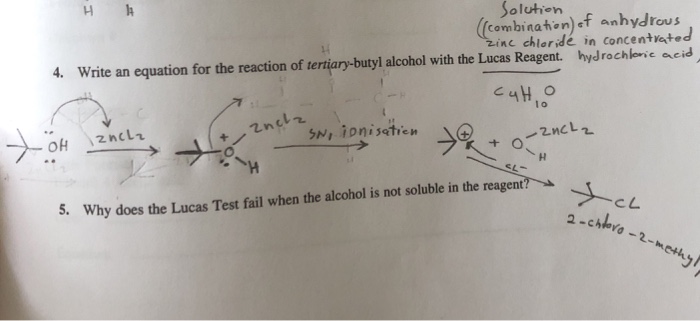As during the reaction a carbocation forms so the rate of. The ZnCl2 coordinates to the hydroxyl oxygen and this generates a far superior leaving group.

What Is The Function Of Zncl2 In Lucas Test Quora
Beim sogenannten Lucas-Test auch Lucas-Probe reagieren die Alkohole abhängig von der Stellung der Hydroxylgruppe im Molekül des Alkohols unterschiedlich schnell oder gar nicht.
Lucas reagent mechanism. Lucas Test Functional Groups. 3 alcohols some but not all 2 alcohols 1 2 3 allylic alcohols Knowns. Da sich der Alkohol vollständig in dem zugegebenen Lucas-Reagenz lösen muss ist der Test.
The Lucas reagent can be prepared by the following steps. Lucasreagent follows SN1 mechanism of reaction because in this test -OH group is protonated by reagent making -OH2 group which further becomes better leaving group. Here you will find curriculum-based online educational resources for Chemistry for all grades.
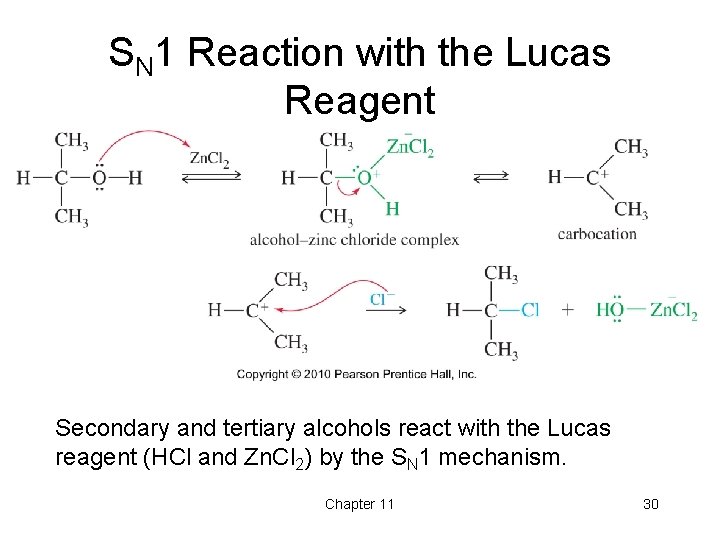
Lucas reagent forms carbocation as intermediate with all three alcohols. Thus when -OH2 group leaves it forms cation and cation stability will be deciding factor of the primary secondary or tertiary alcohol as tertiary alcohol stabilises cation most. It follows the SN1 reaction mechanism.
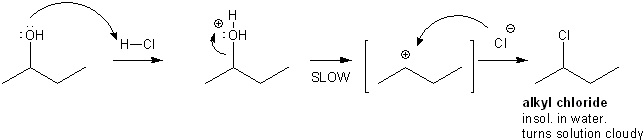
Alcohols can react through an S N 1 mechanism to produce alkyl halides that are insoluble in the aqueous solution and appear as a white precipitate or cloudiness. Reaction of primary secondary and tertiary alcohols with Lucas reagent takes place through unimolecular nucleophilic substitution reaction mechanism. The Lucas reagent concentrated HCl and ZnCl 2 is a test for some alcohols.
Pour the concentrated HCl into a 50 ml graduated cylinder. To prepare lucas reagent you will require concentrated hydrochloric acid HCl and anhydrous zinc chloride ZnCl 2. Place the 100 ml beaker in the ice bath to absorb the heat generated during the dissolution of.
It is used to differentiate primary secondary and tertiary alcohols. Subscribe and get access to thousands of top quality interact. Dabei reagieren Alkohole abhängig von der Stellung der Hydroxygruppe im Molekül des Alkohols unter nucleophiler Substitution unterschiedlich schnell oder gar nicht.
What is the formula of Lucas reagent. The stability of this carbocation differs. The unknown must be soluble in the reagent in order for.
It can be broken down into the following two steps. Measure out 47 ml of concentrated HCl and pour it into the. Lucas reagent offers a polar medium in which S N 1 mechanism is favored.
On adding the alcohol to Lucas reagent a tertiary alcohol reacts immediately forming a ppt of alkyl chloride. The mechanism followed in this reaction is an S N 1 nucleophilic substitution. The reaction of alcohols with Lucas reagent is as follows.
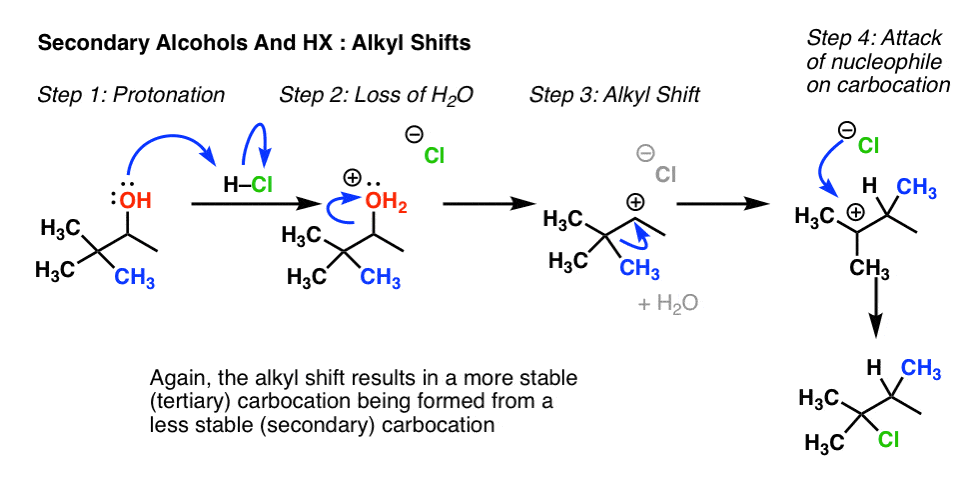
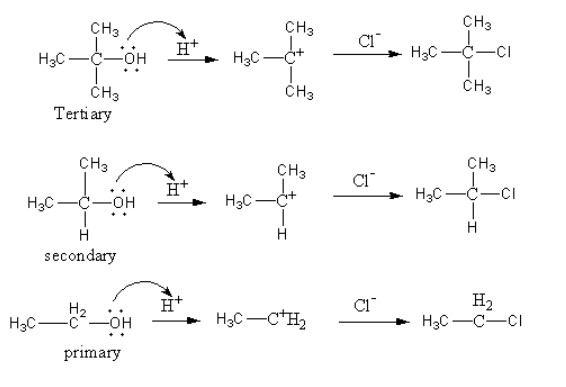
Place the beaker in the ice bath. Das Lucas-Reagenz dient in der organischen Chemie zur Unterscheidung von primären sekundären und tertiären Alkoholen mit niedriger Molarer Masse. In unimolecular nucleophilic substitution the reaction rate is faster when the carbocation intermediate is more stabilized by greater number of electron donating alkyl group R- bonded to the positively charged carbon atom.
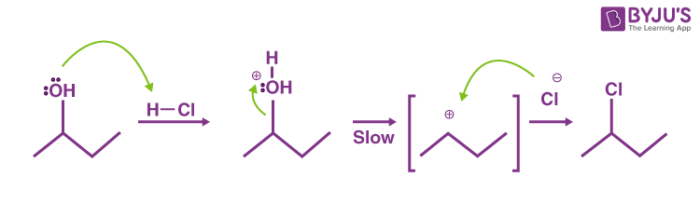
Lucas test is based on the difference in reactivity of alcohols with hydrogen halide. The Lucas test is an important topic of Class XII Chemistry. What happens when secondary alcohol is treated with Lucas reagent.
This organic chemistry video tutorial provides the reaction mechanism of the tollens test which is useful for identifying aldehydes and alpha hydroxy ketones. What is Lucas reagent Class 12. H C l is known as Lucas reagent.
On this carbocation the chloride ion attacks and forms alkyl chloride. Tert-butyl alcohol 3 phenol decene This test is for alcohols with six or fewer carbons. Die Lucas-Probe ist eine Nachweisreaktion in der organischen Chemie zur Unterscheidung von primären sekundären und tertiären Alkoholen.
Lucas Reagent ZnCl2 CH3CH2OH HCl l CH3CH2Cl H2O alcohol hydrogen halide alkyl halide water ZnC 2 This reaction with the Lucas Reagent ZnCl2 is a qualitative test for the different types of alcohols because the rate of the reaction differs greatly for a primary secondary and tertiary alcohol. Primary secondary and tertiary alcohols react with hydrogen halide hydrochloric acid at different rates. This is done to control and absorb the heat which will be generated further.
Measure out 47 ml of concentrated HCl and pour it in a 100 ml beaker. Id say the mechanism of Lucas reagent reaction - for primary alcohol is attack of ZnCl 4 2- or ZnCl 3 - as well as a simple Cl- onto either ROH 2 or ROHZnCl those have similar properties and interconvert one into each other. The OH group belonging to the alcohol is protonated by the hydrochloric acid.
Secondary and tertiary alcohols react via the SN1 mechanism with the Lucas reagent. The mixture of HCl and ZnCl2 is called the Lucas Reagent. The reaction of primary secondary and tertiary alcohols with Lucas reagent takes place via a unimolecular nucleophilic substitution reaction mechanism.
A carbocation is formed as an intermediate with all three types of alcohol.

Why Doesn T Sn2 Reaction Occur With Lucas Reagent With Priamry Alkyl Halide Resulting In Formation Of Alkyl Halide Quora
Organic Chemistry 7 Th Edition L G Wade

Get Answer The Lucas Test Is Used To Check For The Presence The Lucas Test Transtutors
Making Alkyl Halides From Alcohols Master Organic Chemistry
Lucas Test Detailed Mechanism Use In Differentiating Between Different Alcohols
Lucas Reagent Formula Test Preparation Mechanism Msds

Tollens Reagent Mechanism Lucas Chromic Acid Test Youtube
An Unknown Alcohol Is Treated With The Lucas Reagent Class 11 Chemistry Cbse

Analysis Questions 1 Draw The Chemical Equation Or Chegg Com

Test For Alcoholic Group Chemistry Practicals Class 12

Write An Equation For The Reaction Of 2 Butanol With The Lucas Reagent Study Com
Sciencemadness Discussion Board Methanol Hydrochloridric Acid A Dash Of Zinc Chloride Powered By Xmb 1 9 11

2014 Pearson Education Inc Chad Snyder Phd Grace College Chapter 11 Lecture Organic Chemistry 9 Th Edition L G Wade Jr Reactions Of Alcohols Ppt Download

Chem 222 Reaction Of Alcohols With Hcl Youtube
How Do You Identify This As An Sn1 Mechanism I Chegg Com